Developing innovative new ways to treat diseases in people and animals.
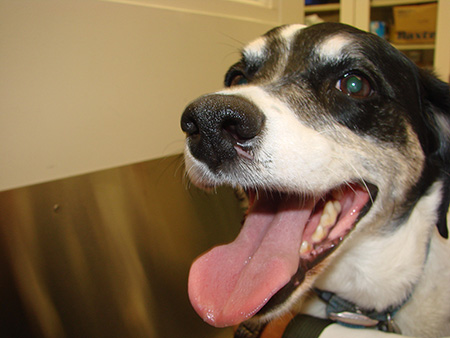
HylaPharm is a university spinout company developing cancer therapies showing promising immune results in real cancers in pet dogs. The strategy is to reverse tumor-induced immune suppression, thus making “cold” tumors “hot” to the immune system. In human patients these aim to increase responsiveness to checkpoint inhibitors, which currently fail in the majority of patients and can cause severe side effects in patients who do respond.
Our team is based at the BTBC incubator in Lawrence Kansas.
2/20/2020: On The Cutting Edge
11/15/2019: HylaPharm completes $700k Kansas Angel Credit raise.
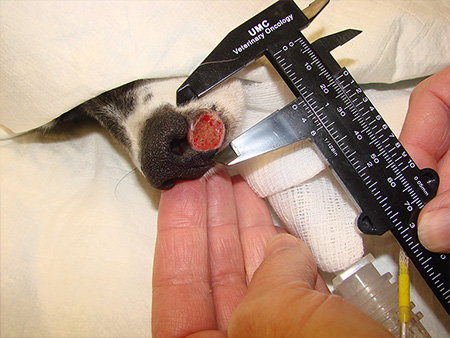
7/28/2019: Pet dogs with mast cell cancer respond to Verisimod.
11/28/2018: HylaPharm completes $750k Kansas Angel Credit raise.
6/8/2018: New treatment from KU Cancer Center, tested on dogs, could eliminate need for chemotherapy.
10/22/2017: HylaPharm is selected to represent the University of Kansas at the Science Coalition’s Sparking Economic Growth conference in Washington D.C.
1/13/2017: HylaPharm has received a second SBIR grant from the National Cancer Institute (division of National Institutes of Health) to develop aerosolized platinum nanoparticle chemotherapy for lung cancer.
1/6/2017: Pet dog with inoperable cancer is now cancer free.

10/1/2016: Hylapharm completed SBIR contract for the National Cancer Institute (division of National Institutes of Health) to develop NanoTor (hyaluronan rapamycin nano conjugates) for treatment of breast cancer. Phase II of this SBIR is pending.